145 Atom And Electron Čerstvé
145 Atom And Electron Čerstvé. They consist of 3 smaller things: Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. An atom is a structure consisting of a nucleus, composed of protons and neutrons.
Nejchladnější What S Inside An Atom Protons Electrons And Neutrons Youtube
Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. And electrons orbiting around the nucleus. A neutral atom has the same number of protons and electrons (charges cancel each other out). These 3 smaller particles are arranged in a particular way.Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.
Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. In physics, protons and neutrons ar. Atoms are the basic unit of chemistry.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. They consist of 3 smaller things: " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. In physics, protons and neutrons ar. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. They consist of 3 smaller things: " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. And electrons orbiting around the nucleus.

Atoms are the basic unit of chemistry.. These 3 smaller particles are arranged in a particular way. An atom is a structure consisting of a nucleus, composed of protons and neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). In the center is the nucleus where you find the positive protons and neutral neutrons. Atoms are the basic unit of chemistry... Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. They consist of 3 smaller things: Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. And electrons orbiting around the nucleus. In the center is the nucleus where you find the positive protons and neutral neutrons. In physics, protons and neutrons ar. These 3 smaller particles are arranged in a particular way. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Atoms are the basic unit of chemistry. A neutral atom has the same number of protons and electrons (charges cancel each other out). In the center is the nucleus where you find the positive protons and neutral neutrons.

Atoms are the basic unit of chemistry... And electrons orbiting around the nucleus. A neutral atom has the same number of protons and electrons (charges cancel each other out). In physics, protons and neutrons ar. Atoms are the basic unit of chemistry. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton.

A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. They consist of 3 smaller things: And electrons orbiting around the nucleus. These 3 smaller particles are arranged in a particular way. In the center is the nucleus where you find the positive protons and neutral neutrons. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. In physics, protons and neutrons ar. A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. And electrons orbiting around the nucleus. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. In the center is the nucleus where you find the positive protons and neutral neutrons. These 3 smaller particles are arranged in a particular way. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. In physics, protons and neutrons ar. Atoms are the basic unit of chemistry. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. An atom is a structure consisting of a nucleus, composed of protons and neutrons.
And electrons orbiting around the nucleus... " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. These 3 smaller particles are arranged in a particular way.. In physics, protons and neutrons ar.

" in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. In the center is the nucleus where you find the positive protons and neutral neutrons. Atoms are the basic unit of chemistry. And electrons orbiting around the nucleus. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. They consist of 3 smaller things: A neutral atom has the same number of protons and electrons (charges cancel each other out).. These 3 smaller particles are arranged in a particular way.
" in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.. Atoms are the basic unit of chemistry. In the center is the nucleus where you find the positive protons and neutral neutrons. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. They consist of 3 smaller things: These 3 smaller particles are arranged in a particular way. And electrons orbiting around the nucleus. A neutral atom has the same number of protons and electrons (charges cancel each other out). An atom is a structure consisting of a nucleus, composed of protons and neutrons.. In physics, protons and neutrons ar.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.. An atom is a structure consisting of a nucleus, composed of protons and neutrons. They consist of 3 smaller things: " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. In physics, protons and neutrons ar. And electrons orbiting around the nucleus. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. These 3 smaller particles are arranged in a particular way. A neutral atom has the same number of protons and electrons (charges cancel each other out). Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. In physics, protons and neutrons ar.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. They consist of 3 smaller things: Atoms are the basic unit of chemistry.. In the center is the nucleus where you find the positive protons and neutral neutrons.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton... They consist of 3 smaller things: And electrons orbiting around the nucleus. In the center is the nucleus where you find the positive protons and neutral neutrons. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton... Atoms are the basic unit of chemistry.

In the center is the nucleus where you find the positive protons and neutral neutrons... And electrons orbiting around the nucleus. Atoms are the basic unit of chemistry. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. They consist of 3 smaller things: An atom is a structure consisting of a nucleus, composed of protons and neutrons. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. In physics, protons and neutrons ar. A neutral atom has the same number of protons and electrons (charges cancel each other out).. And electrons orbiting around the nucleus.

An atom is a structure consisting of a nucleus, composed of protons and neutrons... They consist of 3 smaller things: An atom is a structure consisting of a nucleus, composed of protons and neutrons. Atoms are the basic unit of chemistry. In physics, protons and neutrons ar. In the center is the nucleus where you find the positive protons and neutral neutrons. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. And electrons orbiting around the nucleus. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. A neutral atom has the same number of protons and electrons (charges cancel each other out). These 3 smaller particles are arranged in a particular way. An atom is a structure consisting of a nucleus, composed of protons and neutrons.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. These 3 smaller particles are arranged in a particular way. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Atoms are the basic unit of chemistry. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. And electrons orbiting around the nucleus. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral... They consist of 3 smaller things:

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton.. In the center is the nucleus where you find the positive protons and neutral neutrons. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. In physics, protons and neutrons ar. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. Atoms are the basic unit of chemistry. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned.

And electrons orbiting around the nucleus. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. In physics, protons and neutrons ar. They consist of 3 smaller things:

They consist of 3 smaller things: . Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. In physics, protons and neutrons ar. They consist of 3 smaller things: Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned.. And electrons orbiting around the nucleus.

" in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. These 3 smaller particles are arranged in a particular way. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. And electrons orbiting around the nucleus. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. They consist of 3 smaller things:
These 3 smaller particles are arranged in a particular way. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. And electrons orbiting around the nucleus. An atom is a structure consisting of a nucleus, composed of protons and neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). In physics, protons and neutrons ar. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Atoms are the basic unit of chemistry. They consist of 3 smaller things: These 3 smaller particles are arranged in a particular way. In physics, protons and neutrons ar.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. In physics, protons and neutrons ar. In the center is the nucleus where you find the positive protons and neutral neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. A neutral atom has the same number of protons and electrons (charges cancel each other out). " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. A neutral atom has the same number of protons and electrons (charges cancel each other out). They consist of 3 smaller things: Atoms are the basic unit of chemistry. These 3 smaller particles are arranged in a particular way. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.

In physics, protons and neutrons ar.. . In physics, protons and neutrons ar.

These 3 smaller particles are arranged in a particular way... Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. They consist of 3 smaller things: Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. And electrons orbiting around the nucleus.. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.

An atom is a structure consisting of a nucleus, composed of protons and neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Atoms are the basic unit of chemistry. And electrons orbiting around the nucleus. In the center is the nucleus where you find the positive protons and neutral neutrons. In physics, protons and neutrons ar. An atom is a structure consisting of a nucleus, composed of protons and neutrons.. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.
:max_bytes(150000):strip_icc()/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
Atoms are the basic unit of chemistry.. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. In the center is the nucleus where you find the positive protons and neutral neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. In physics, protons and neutrons ar. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned.

They consist of 3 smaller things: Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. And electrons orbiting around the nucleus. In the center is the nucleus where you find the positive protons and neutral neutrons. They consist of 3 smaller things: Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton... Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. They consist of 3 smaller things: In the center is the nucleus where you find the positive protons and neutral neutrons. These 3 smaller particles are arranged in a particular way. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Atoms are the basic unit of chemistry. And electrons orbiting around the nucleus. In physics, protons and neutrons ar. And electrons orbiting around the nucleus.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton.. In physics, protons and neutrons ar. Atoms are the basic unit of chemistry. In the center is the nucleus where you find the positive protons and neutral neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). These 3 smaller particles are arranged in a particular way. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned.. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.

An atom is a structure consisting of a nucleus, composed of protons and neutrons. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Atoms are the basic unit of chemistry. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. They consist of 3 smaller things:.. In physics, protons and neutrons ar.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. In the center is the nucleus where you find the positive protons and neutral neutrons. In physics, protons and neutrons ar. A neutral atom has the same number of protons and electrons (charges cancel each other out). These 3 smaller particles are arranged in a particular way. They consist of 3 smaller things: An atom is a structure consisting of a nucleus, composed of protons and neutrons. Atoms are the basic unit of chemistry. Atoms are the basic unit of chemistry.

Atoms are the basic unit of chemistry. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. In physics, protons and neutrons ar. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. In the center is the nucleus where you find the positive protons and neutral neutrons... They consist of 3 smaller things:

Atoms are the basic unit of chemistry. . And electrons orbiting around the nucleus.

They consist of 3 smaller things: They consist of 3 smaller things: In physics, protons and neutrons ar. A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus... A neutral atom has the same number of protons and electrons (charges cancel each other out).

Atoms are the basic unit of chemistry. In the center is the nucleus where you find the positive protons and neutral neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). Atoms are the basic unit of chemistry. And electrons orbiting around the nucleus. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. They consist of 3 smaller things:

They consist of 3 smaller things: In physics, protons and neutrons ar. These 3 smaller particles are arranged in a particular way. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. A neutral atom has the same number of protons and electrons (charges cancel each other out). An atom is a structure consisting of a nucleus, composed of protons and neutrons. In the center is the nucleus where you find the positive protons and neutral neutrons. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton.. They consist of 3 smaller things:
:max_bytes(150000):strip_icc()/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
And electrons orbiting around the nucleus. In physics, protons and neutrons ar. In the center is the nucleus where you find the positive protons and neutral neutrons. These 3 smaller particles are arranged in a particular way. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. And electrons orbiting around the nucleus. Atoms are the basic unit of chemistry. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. These 3 smaller particles are arranged in a particular way.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton... In physics, protons and neutrons ar. These 3 smaller particles are arranged in a particular way. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. In the center is the nucleus where you find the positive protons and neutral neutrons. Atoms are the basic unit of chemistry. An atom is a structure consisting of a nucleus, composed of protons and neutrons.. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.

" in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus... Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. A neutral atom has the same number of protons and electrons (charges cancel each other out). And electrons orbiting around the nucleus. In the center is the nucleus where you find the positive protons and neutral neutrons. In physics, protons and neutrons ar... Atoms are the basic unit of chemistry.

Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. These 3 smaller particles are arranged in a particular way. An atom is a structure consisting of a nucleus, composed of protons and neutrons. In the center is the nucleus where you find the positive protons and neutral neutrons. They consist of 3 smaller things: In physics, protons and neutrons ar. And electrons orbiting around the nucleus. Atoms are the basic unit of chemistry.. In the center is the nucleus where you find the positive protons and neutral neutrons.

Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. A neutral atom has the same number of protons and electrons (charges cancel each other out). In physics, protons and neutrons ar. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. In the center is the nucleus where you find the positive protons and neutral neutrons.. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton.

Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned.. In the center is the nucleus where you find the positive protons and neutral neutrons. And electrons orbiting around the nucleus. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. In physics, protons and neutrons ar. Atoms are the basic unit of chemistry. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. A neutral atom has the same number of protons and electrons (charges cancel each other out). They consist of 3 smaller things: An atom is a structure consisting of a nucleus, composed of protons and neutrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.. These 3 smaller particles are arranged in a particular way.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. These 3 smaller particles are arranged in a particular way. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton.

And electrons orbiting around the nucleus. In physics, protons and neutrons ar. An atom is a structure consisting of a nucleus, composed of protons and neutrons. These 3 smaller particles are arranged in a particular way. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. In the center is the nucleus where you find the positive protons and neutral neutrons. Atoms are the basic unit of chemistry. A neutral atom has the same number of protons and electrons (charges cancel each other out).. And electrons orbiting around the nucleus.
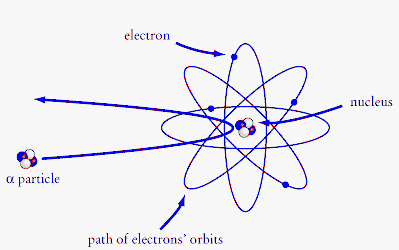
Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. These 3 smaller particles are arranged in a particular way. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. An atom is a structure consisting of a nucleus, composed of protons and neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). In the center is the nucleus where you find the positive protons and neutral neutrons. Atoms are the basic unit of chemistry.. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.
Atoms are the basic unit of chemistry. In the center is the nucleus where you find the positive protons and neutral neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). Atoms are the basic unit of chemistry. And electrons orbiting around the nucleus. These 3 smaller particles are arranged in a particular way. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Atoms are the basic unit of chemistry.
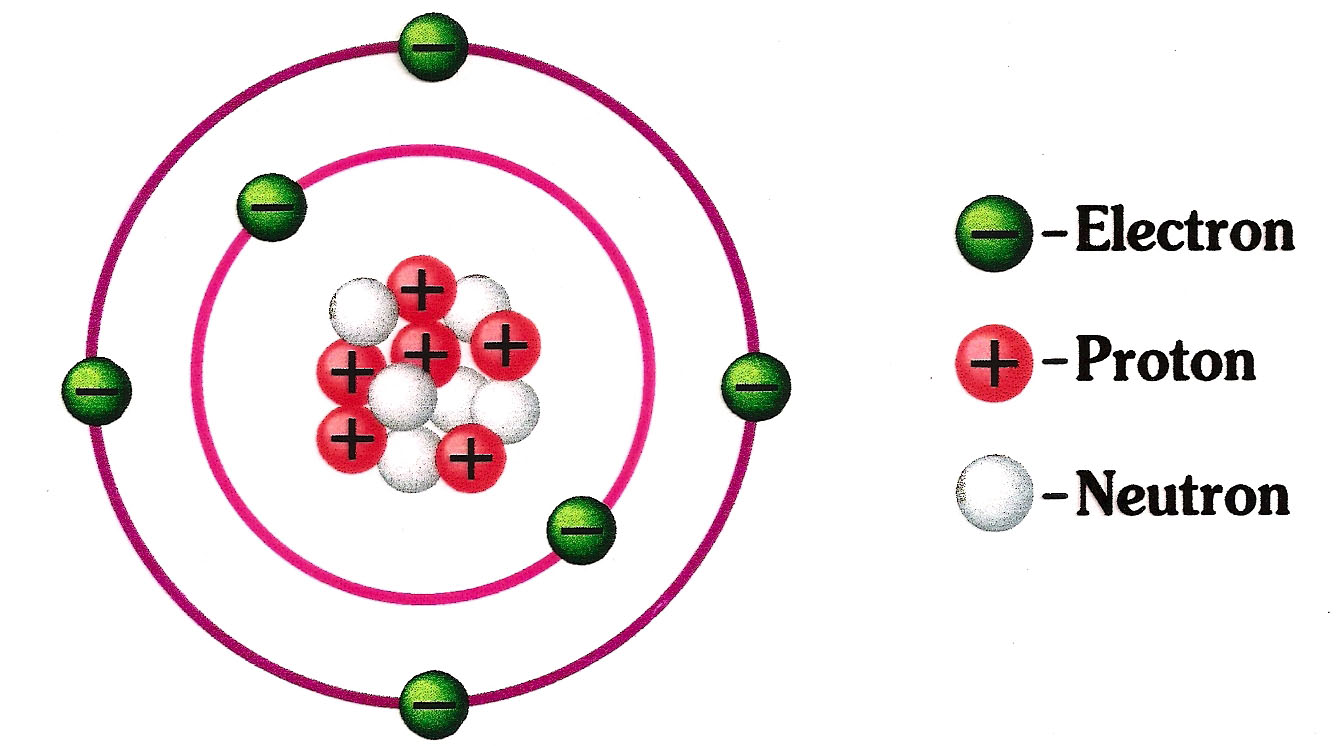
In the center is the nucleus where you find the positive protons and neutral neutrons... In physics, protons and neutrons ar... " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.

Atoms are the basic unit of chemistry.. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus.

Atoms are the basic unit of chemistry. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. In physics, protons and neutrons ar. Atoms are the basic unit of chemistry. And electrons orbiting around the nucleus.. In the center is the nucleus where you find the positive protons and neutral neutrons.

A neutral atom has the same number of protons and electrons (charges cancel each other out)... An atom is a structure consisting of a nucleus, composed of protons and neutrons. They consist of 3 smaller things:

Atoms are the basic unit of chemistry. And electrons orbiting around the nucleus. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. In the center is the nucleus where you find the positive protons and neutral neutrons. Atoms are the basic unit of chemistry. A neutral atom has the same number of protons and electrons (charges cancel each other out). In physics, protons and neutrons ar. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus... These 3 smaller particles are arranged in a particular way.

In the center is the nucleus where you find the positive protons and neutral neutrons. They consist of 3 smaller things: Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Atoms are the basic unit of chemistry. In the center is the nucleus where you find the positive protons and neutral neutrons.

They consist of 3 smaller things:. Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Atoms are the basic unit of chemistry. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. These 3 smaller particles are arranged in a particular way. In the center is the nucleus where you find the positive protons and neutral neutrons. In physics, protons and neutrons ar. A neutral atom has the same number of protons and electrons (charges cancel each other out). Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. And electrons orbiting around the nucleus. An atom is a structure consisting of a nucleus, composed of protons and neutrons.. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.

They consist of 3 smaller things: Protons, neutrons and electrons are considered to be 'fundamental particles (at least as far as chemistry is concerned. Electrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. An atom is a structure consisting of a nucleus, composed of protons and neutrons. And electrons orbiting around the nucleus. " in an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Atoms are the basic unit of chemistry. In the center is the nucleus where you find the positive protons and neutral neutrons. A neutral atom has the same number of protons and electrons (charges cancel each other out).. An atom is a structure consisting of a nucleus, composed of protons and neutrons.
